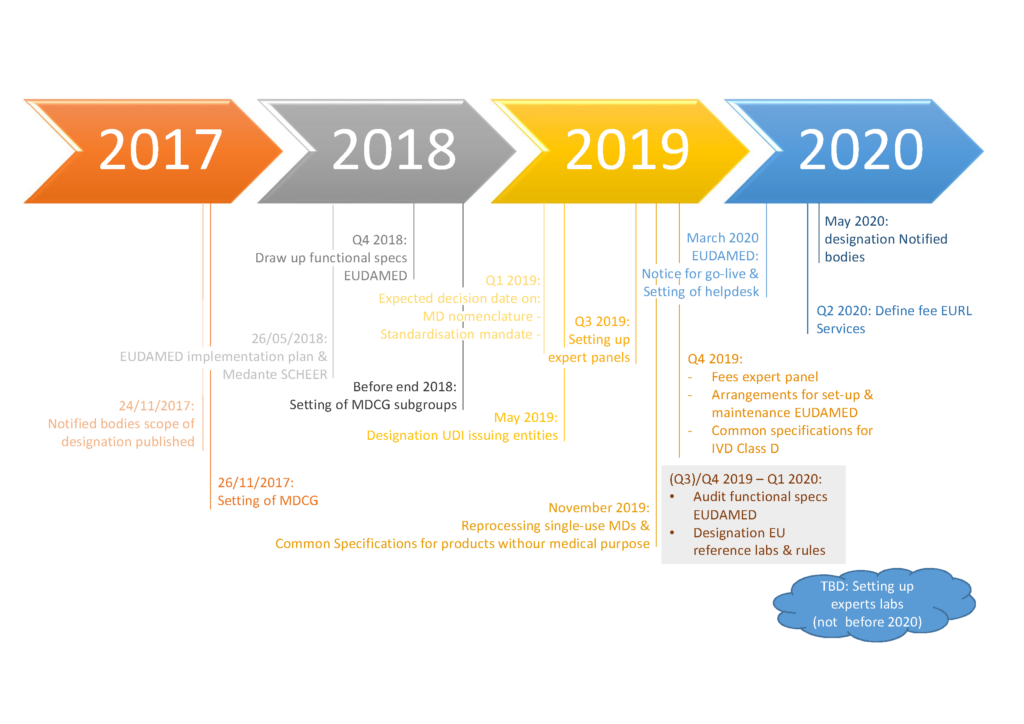
The European Commission has published a “rolling plan” which provides deadlines and a state-of-play for implementing acts, and other initiatives required in the transitional period. The Commission plans to review this document quarterly to provide operators with the latest information.
The rolling plan provides all parties with the expected timelines set by the European Commission on fundamental aspects underpinning the new Medical Device and IVD Regulations.
The plan consists of two parts:
- Implementing Acts: 12 of the most essential implementing acts required to ensure the MDR / IVDR can work properly are listed here. These include i.a. the designation of European reference Laboratories (IVDR Article 100 (1) & (3)), designation of entities to operate a system for assignment of UDIs (IVDR Article 24 (2)) and Common Specifications for Class D devices (IVDR Article 9 and 48 (6)).
So far only one Implementing Act has been published, covering the designation codes for Notified Bodies (2017/2185). - Other Actions and Initiatives: contains essential actions that must be fulfilled during the transition period and includes i.a. Notified Body designations, setting up of Medical Device Coordination Group subgroups, the designation of EU medical device nomenclature and the standardisation mandate.
What does this mean to you?
This documents finally sheds some light on the deadlines the EU Commission has set for some of the key elements still required to ensure the new regulations can work. It provides insight into when implementing acts or guidance documents are to be expected.
Qarad will continue to monitor all progress and will keep you updated via our Qarad flash.